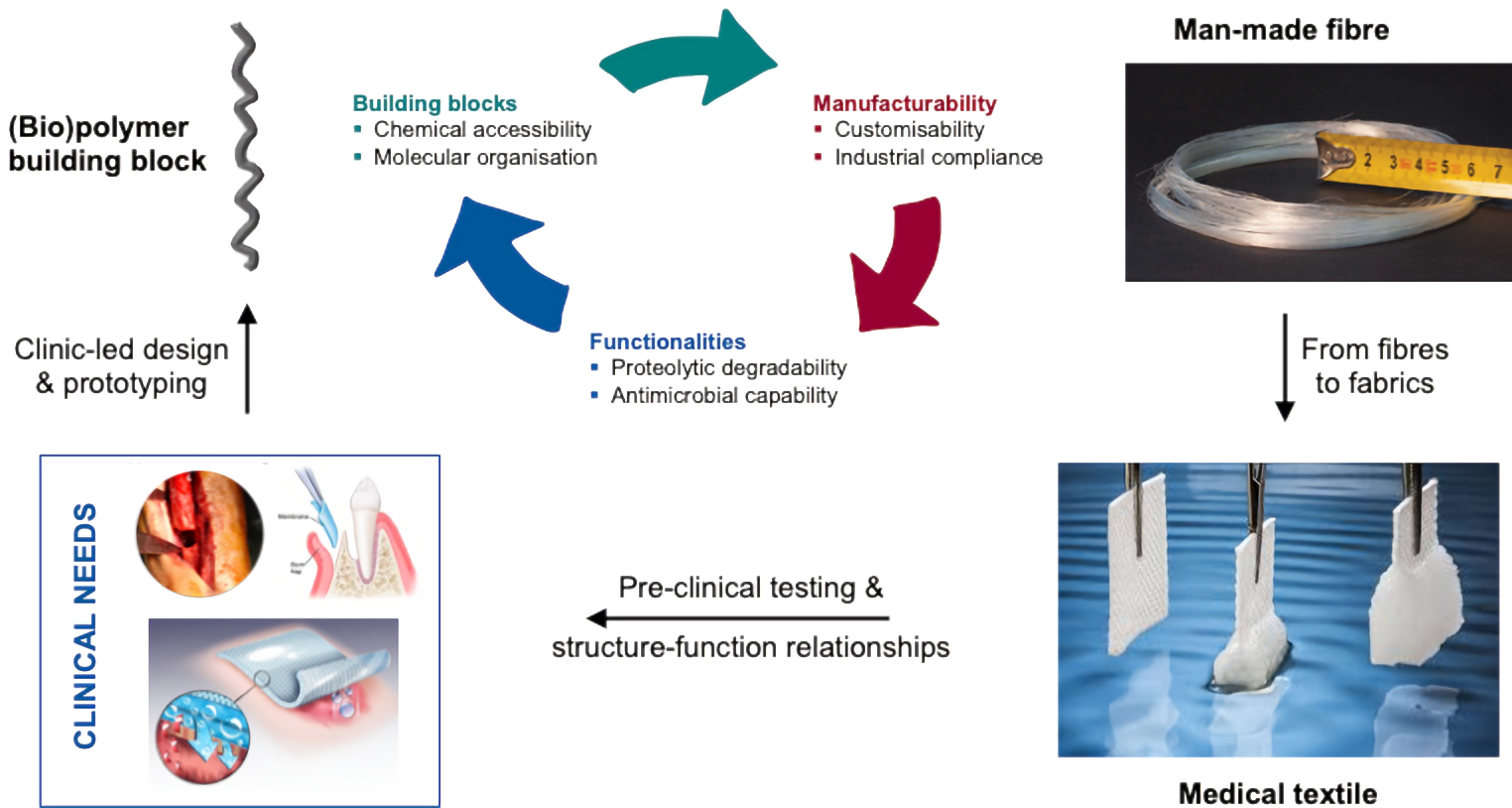
Research Activities

Research in the CCTMIH began by addressing unmet needs in wound management, continence management, filtration and infection control. As the research has evolved in response to clinical challenges, the development of new implantable and bioactive textile materials has become a major focus, aimed at addressing performance gaps in tissue repair and regeneration, diagnostics, wound healing and the treatment of disease. This work involves research on biomaterials such as collagen, textile manufacturing and medical device design, as well as clinical evaluation.
Advanced wound management and wound theranostics
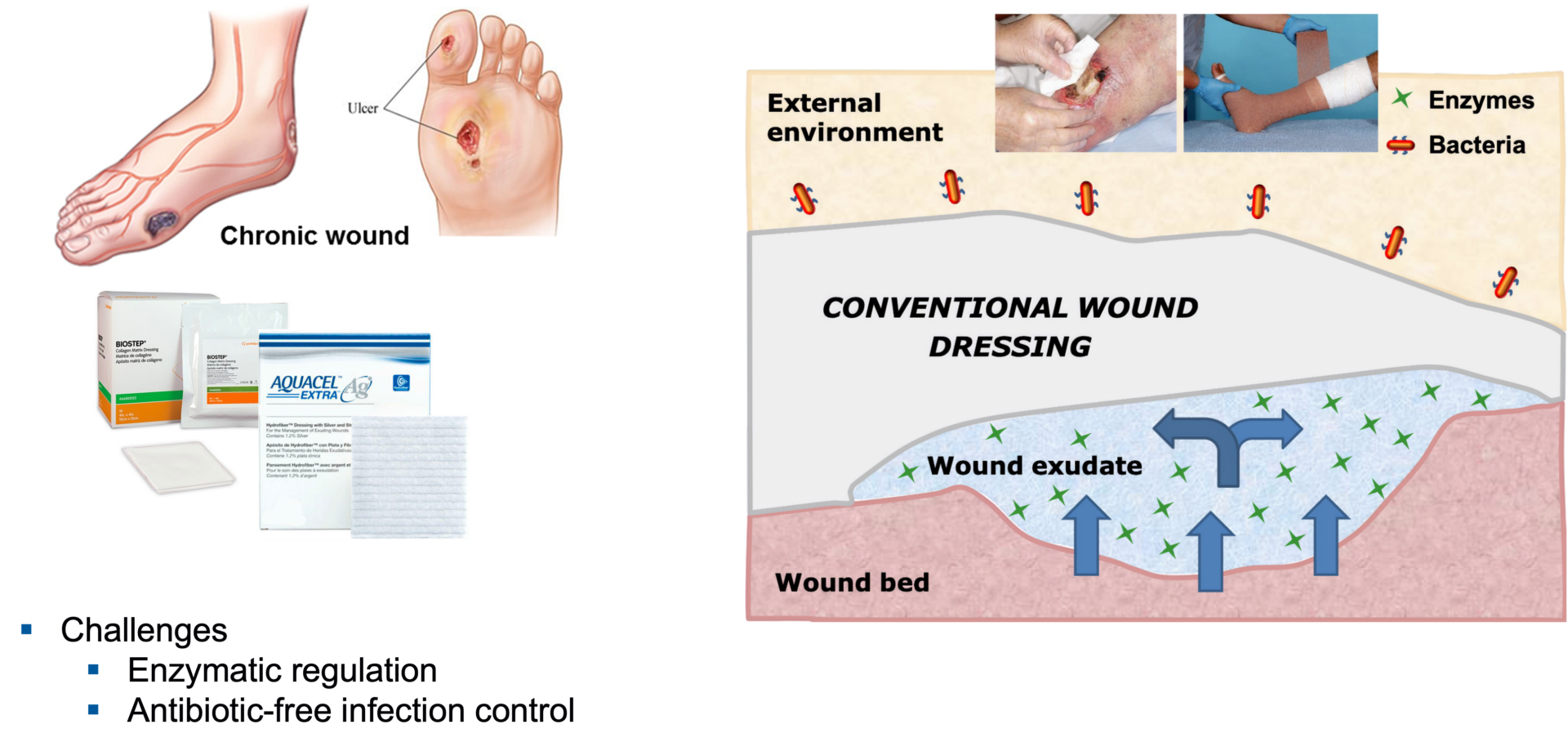
Chronic wounds can arise for reasons such as poor circulation, neuropathy, difficulty moving, systemic illnesses, age, and repeated trauma. Chronic wounds can include venous leg ulcers, pressure sores and burn injuries.
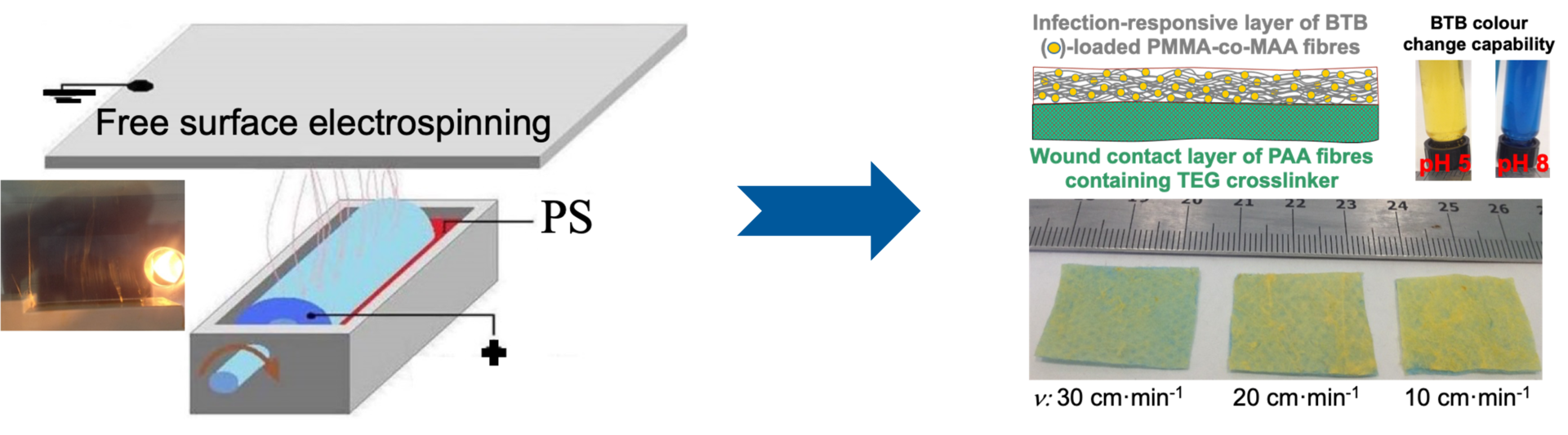
Advanced wound dressings capable of managing infection, enzymatic activity, and exudate are areas of development. Research strategies include photodynamically active fibres, MMP-chelating collagen dressings as well as infection-responsive materials for improved wound diagnosis and treatment. The build-up of complex multilayered construct is recently being pursued as a new wound theranostic approach, thanks to a recent grant from the Clothworkers company.
With increasing rates of diabetes and obesity, significantly more people will suffer from chronic wounds that will not heal quickly. Chronic wounds cause severe emotional and physical stress as well as creating a significant financial burden on patients and the entire healthcare system.
Novel material processing technologies for soft robotic surgery
Pancreatic cancer is currently the deadliest form of cancer (mortality 95% <2yrs). Through a multidisciplinary EPSRC-sponsored network of engineers, clinicians and industrial partners, we are investigating new minimally invasive strategies for the diagnostics and treatment of pancreatic cancer. We aim to develop elastomeric material systems that respond to multiple externally-activated stimuli to accomplish non-destructive access to the pancreas' intricate internal anatomy, as well as localised delivery of drugs within the pancreas. This will be investigated by a research team of academics, Research Fellows and PhD researchers working at the interface of manufacturing technology, robotics, and materials processing.
Long-lasting barrier membranes for guided bone regeneration
Resorbable collagen membranes are a key component of guided bone regeneration (GBR) therapy, supporting the regrowth of hard tissue cells and preventing the infiltration of soft tissue cells.
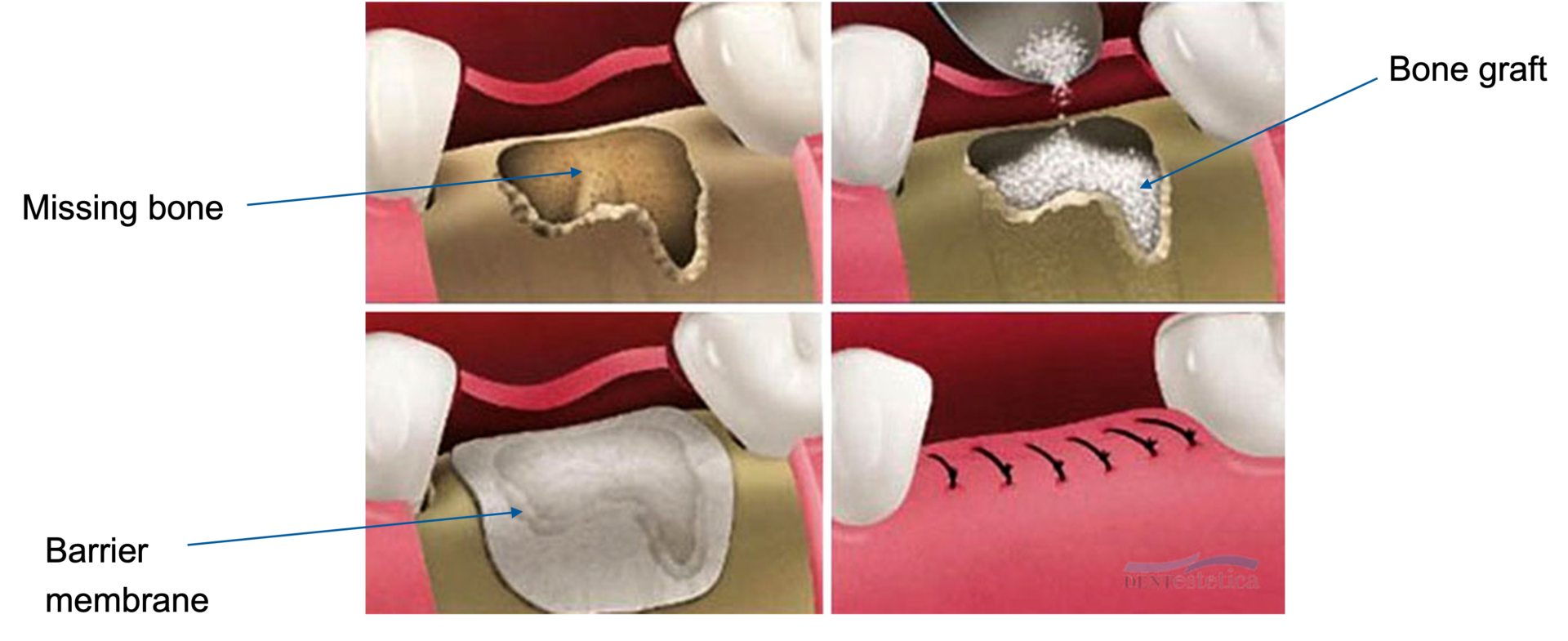
Existing commercial collagen membranes have been documented to suffer from restricted enzymatic stability, raising risks of poor regenerated bone quality, underperforming clinical outcomes, and need of multiple surgeries for patients. Working with dental and orthopaedic surgeons, we are addressing these challenges by targeting the molecular scale of collagen, aiming to introduce photoinduced covalent crosslinks of varying molecular rigidity. By converting these synthetic products into macroscopic membrane prototypes, we can accomplish longer-lasting proteolytic stability, as well as controlled swelling ratio and compression properties, as key properties for successful GBR therapy. Our collaborations with Leeds School of Dentistry enable us to pursue prototype testing in vitro and in vivo, prior to moving towards first-in-human clinical evaluation.
Infection Control and Healthcare Associated Infections
(HCAIs) are major global concerns. Research involves developing alternative, antibiotic-free strategies (e.g. photodynamic therapy fibres) for the reduction of surgical and wound site infections, more effective removal of pathogens from surfaces, as well as the early-stage sensing and signalling of wound infection. Management of microbial activity, particularly bioflims in wound sites is key to reducing healing times, and new multi-functional textile materials capable of preventing biofilm formation are in development.

Development of Leeds collagen technology - HyFaCol
Collagen has been widely applied in biomedical applications, including as a wound dressing material, barrier membrane for guided bone regeneration, as well as a joint injection for osteoarthritis management. However, its limited mechanical properties and processability prevent the development of personalised collagen products that meet patient and end user needs. At Leeds, we have leveraged our multidisciplinary expertise in biopolymer engineering, fibre manufacture and clinical translation to accomplish a Hydrogel of Functionalised ateloCollagen (HyFaCol) technology that has been patented in the US and Europe. HyFaCol enables varying profiles of macroscopic properties and format to be accomplished in order to meet the requirements of complex healthcare applications.
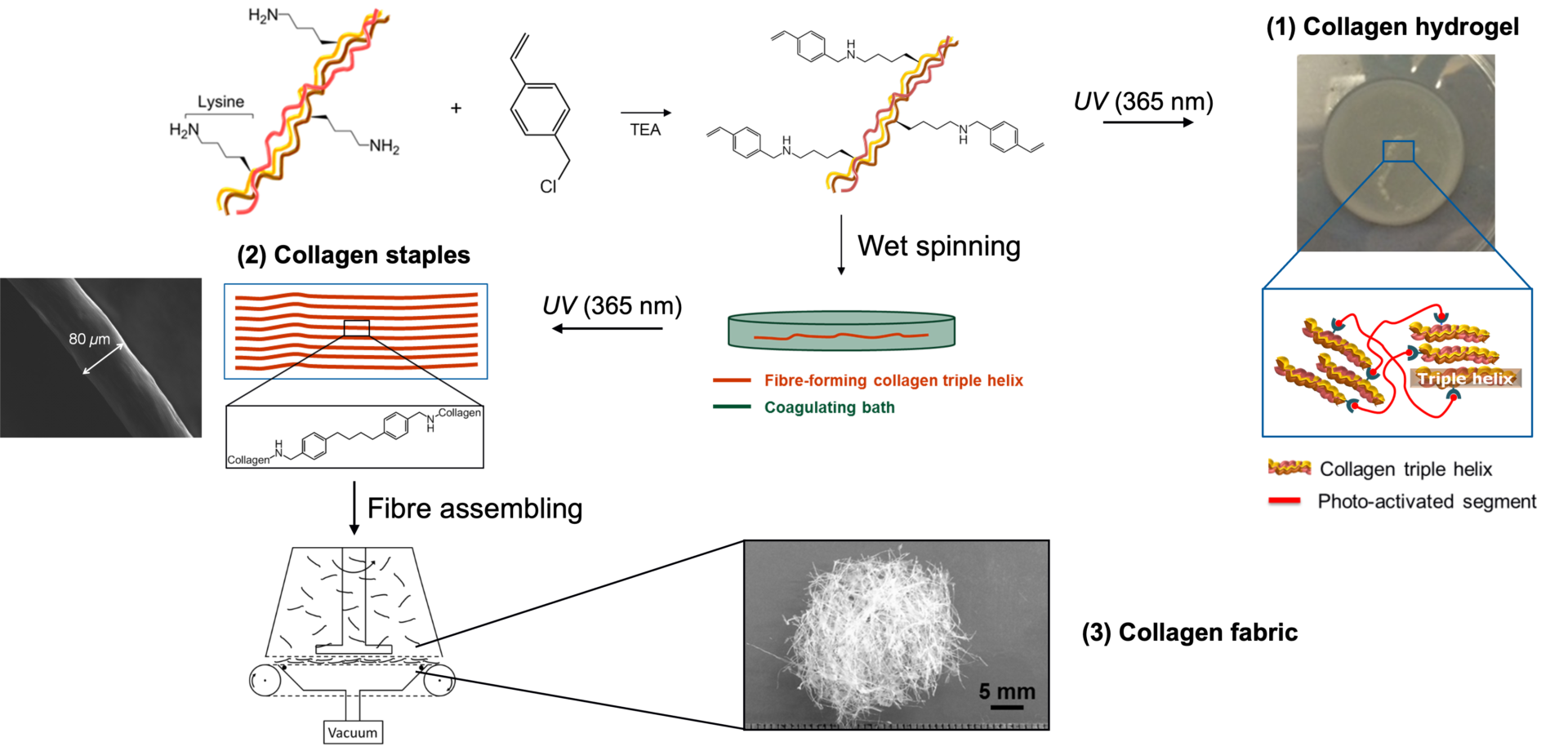
The technology has been successfully evaluated in vitro and in vivo, and we are now working to secure regulatory approval for a first-in-human clinical evaluation (clinical lead: Dr. Francesco del Galdo, Leeds Chapel Allerton Hospital).
Continence management
Our work aims to significantly increase device disposal options for patients, carers and NHS staff by developing technology capable of enabling discrete, leakage resistant, fully toilet flushable devices. Morbidity, social withdrawal and nursing time issues are also being tackled by developing alternative technologies for combatting odour, reducing bacterial activity, skin irritation and the risk of infection associated with extended use of ostomy and ileostomy products.
Management of Disease
Sophisticated fibre-based and injectable devices are required to target the delivery of therapeutic compounds to diseased soft and hard tissues, as well as to enable improved therapeutic treatments for patients at the bed-side to manage pain or disease.